Mass Spectrometry
Mass spectrometry (MS) is an analytical method that utilizes the mass-to-charge (m/z) ratio to identify compounds present in a sample. By determining the molecular weight and analyzing isotopic abundance, this technique identifies compounds accurately. The process involves ionizing the sample into gaseous ions and then analyzing them based on their mass-to-charge ratios and relative abundances.
In modern times, mass spectrometry has become a well-established and highly advantageous detection method due to its selectivity, sensitivity, and capability for multi-sample analysis. It can be integrated with various chromatographic techniques, including liquid chromatography, thin layer chromatography, gas chromatography, and inductively coupled plasma. As a result, mass spectrometry finds extensive use across multiple research fields and industries, including pharmaceuticals, food, health clinics, clinical research labs, and forensic and environmental testing labs.
HOW DOES MASS SPECTROMETRY WORK?
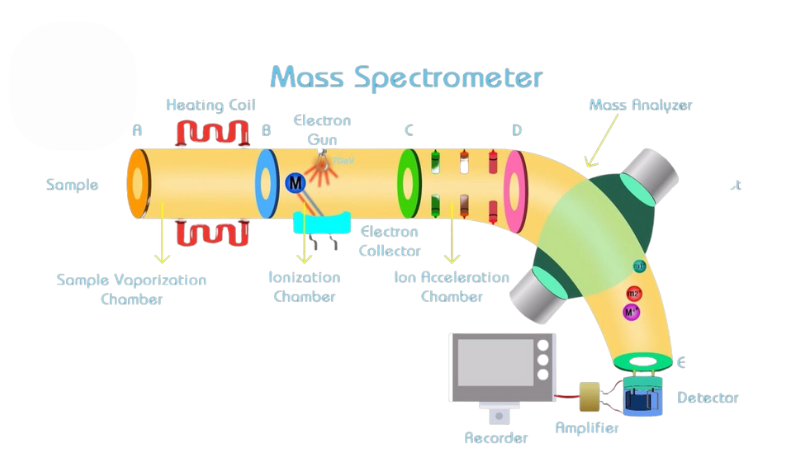
A mass spectrometer operates by transforming individual molecules into ions and analyzing the resulting ions’ relative abundance. Inside the ion chamber, each molecule is ionized, forming a molecular ion with one less electron than the parent molecule. These molecular ions, also called ‘radical cations,’ undergo successive fragmentation into more ions, creating a variety of ions from a complex sample. The ions are then accelerated in an electromagnetic field and separated based on their mass-to-charge (m/z) ratios. The detector in the instrument records the ions in proportion to their relative abundance, producing a mass spectrum of the molecule.
APPLICATIONS OF MASS SPECTROMETRY
Mass spectrometry is extensively utilized due to its sensitivity, enabling the measurement of very low molecular weights at extremely low concentrations, below nanograms per milliliter (ng/mL). Its versatility is further enhanced by the ability to combine mass spectrometry with other separation techniques like capillary electrophoresis, GC, and HPLC, enabling simultaneous separation and identification of analytes.
Some common applications of mass spectrometry include:
– Analyzing amino acid sequences of proteins and peptides
– Evaluating impurities during drug development
– Assessing the purity of active pharmaceutical ingredients
– Routine analysis of illegal drugs in urine, blood, and hair
– Detecting hereditary disorders related to amino acid, fatty acid, and organic biosynthesis.